Prostate Cancer Resources for Providers
Below is a library of article, studies, videos and CME courses about prostate cancer screening and biomarker tests. These resources are intended for primary care and urology providers.
Articles
PCMarkers.com articles for providers.

A Guide to PSA Testing for Primary Care Providers
PCPs play crucial role in reducing late-stage prostate cancer diagnoses.

Reinstating PSA Testing
While routine PSA testing has significantly declined since 2008, the incidence of metastatic prostate cancer has increased for men 45 and older across all races, ethnicities, and geographic locations in the US. Those trends plus the development of prostate cancer biomarkers to help prevent unnecessary biopsies and treatment support the return of routine PSA testing.
%s
Provider Resources (Printable)
Below are resources providers can use in their practice to help guide prostate cancer screening decisions. We also have provided a patient flyer that will help you communicate important reasons for prostate cancer screening.
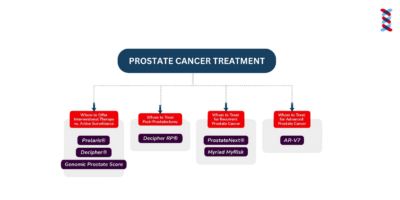
Phases of Prostate Cancer
Learn which prostate cancer biomarkers to use at various stages from screening to advanced cancer.

PCM Test Chart
The chart breaks down all PCM tests to display who they are targeted for and what results and decisions they provide.
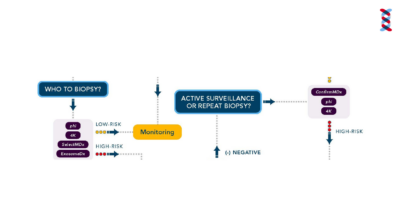
Prostate Cancer Algorithm
This algorithm helps providers understand the next steps from initial screening through treatment.

Patient's "Know Your Risk" Checklist
This flyer is meant for patients to print and use as a tool to assess their own risk, before visiting and talking through with their provider.
Video for PCPs
Rethinking Prostate Cancer Screening: How Risk Stratification Saves Lives
Rethinking Prostate Cancer Screening: Video for PCPs
Learn how risk-stratified PSA testing helps primary care providers detect prostate cancer early while avoiding unnecessary biopsies. Watch now for smarter screening guidelines.
%s
Prostate Cancer Studies
Peer-reviewed journal articles about prostate cancer screening and testing.
A multi-institutional prospective trial in the USA confirms that the 4Kscore accurately identifies men with high-grade prostate cancer
The 4Kscore combines measurement of four kallikreins in blood with clinical information as a measure of the probability of significant (Gleason ≥7) prostate cancer (PCa) before prostate biopsy. In this study, the 4Kscore showed excellent diagnostic performance in detecting significant PCa. It is a useful tool in selecting men who have significant disease and are […]
Beyond PSA: The Role of Prostate Health Index (phi)
Literature data showed that phi had good diagnostic performance to identify clinically significant (cs) PCa, suggesting that it could be a useful tool for personalized treatment decision-making. In this review, phi potentialities, limitations, and comparisons with other blood- and urinary-based tests were explored.
Commercialized Blood-, Urinary- and Tissue-Based Biomarker Tests for Prostate Cancer Diagnosis and Prognosis
In the diagnosis and prognosis of prostate cancer (PCa), the serum prostate-specific antigen test is widely used but is associated with low specificity. Therefore, blood-, urinary- and tissue-based biomarker tests have been developed, intended to be used in the diagnostic and prognostic setting of PCa. This review provides an overview of commercially available biomarker tests […]
Detection of High-grade Prostate Cancer Using a Urinary Molecular Biomarker-Based Risk Score
The risk score based on the mRNA liquid biopsy assay combined with traditional clinical risk factors identified men at risk of harboring high-grade PCa and resulted in a better patient risk stratification compared with current methods in clinical practice. Therefore, the risk score could reduce the number of unnecessary prostate biopsies.
Pre-diagnosis urine exosomal RNA (ExoDx EPI score) is associated with post-prostatectomy pathology outcome
ExoDx Prostate IntelliScore (EPI) is a non-invasive urine exosome RNA-based test for risk assessment of high-grade prostate cancer. We evaluated the association of pre-biopsy test results with post-radical prostatectomy (RP) outcomes to understand the potential utility of EPI to inform invasive treatment vs active surveillance (AS) decisions.
Predicting high-grade prostate cancer at initial biopsy: clinical performance of the ExoDx (EPI) Prostate Intelliscore test in three independent prospective studies
The ExoDx Prostate (IntelliScore) (EPI) test stratifies patients for risk of high-grade prostate cancer (HGPC; ≥ Grade Group 2 [GG] PC) in men ≥ 50 years with equivocal prostate-specific antigen (PSA) (2–10 ng/mL). Here, we present a pooled meta-analysis from three independent prospective-validation studies in men presenting for initial biopsy decision.
Prospective study of diagnostic accuracy in the detection of high-grade prostate cancer in biopsy-naïve patients with clinical suspicion of prostate cancer who underwent the Select MDx test
This study aimed to externally validate the diagnostic accuracy of the Select MDx test for Significant prostate cancer (Sig PCa) (ISUP > 1), in a contemporaneous, prospective, multicenter cohort with a prostate-specific antigen (PSA) between 3 and 10 ng/ml and a non-suspicious digital rectal examination.
Prostate Health Index and Prostate Health Index Density as Diagnostic Tools for Improved Prostate Cancer Detection
The objective of the present study was to evaluate the diagnostic potential of p2PSA, %p2PSA, phi, and phi density (PHID) as independent biomarkers and in combination with other demographic and clinical parameters, to predict overall and clinically significant PCa. The ability of p2PSA and its derivatives to discriminate HGPIN at biopsy was evaluated.
The prostate health index selectively identifies clinically significant prostate cancer
We investigate whether phi improves specificity for detecting clinically significant prostate cancer and can help reduce prostate cancer over diagnosis. The test outperforms its individual components of total, free and [-2]proPSA for the identification of clinically significant prostate cancer. Phi may be useful as part of a multivariable approach to reduce prostate biopsies and over […]
The Role of Genetic Testing in Prostate Cancer
A. Karim Kader, MD, PhD, FRCSC, Professor in the Department of Urology and Director of Urologic Oncology at the University of California, San Diego, argues that genomic risk stratification plays an important role in our understanding of prostate cancer. He highlights the differences between germline and somatic genomic classifiers and the impact they can have on prostate […]
Assessment of the Validity of Nuclear-Localized Androgen Receptor Splice Variant 7 in Circulating Tumor Cells as a Predictive Biomarker for Castration-Resistant Prostate Cancer
This study of 142 men with metastatic castration-resistant prostate cancer (mCRPC) found that nuclear-localized AR-V7 protein in circulating tumor cells can be an indication of whether or not these patients have better survival rates with taxane chemotherapy compared to androgen receptor signaling (ARS)-directed therapy.
96% Negative Predictive Value for High-grade Cancer
This study shows that, when compared to other risk factors, detection of DNA-methylation in histopathologically negative biopsies was the most significant and important predictor of high-grade cancer, resulting in a negative predictive value of 96%.
A 17-gene Panel for Prediction of Adverse Prostate Cancer Pathologic Feature
This real-world multi-institutional study found the Genomic Prostate Score Test is an effective, independent predictor of adverse pathology at surgery and provided patients with greater confidence and assurance in their decision.
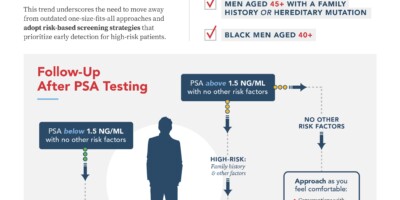
An Approach Using PSA Levels of 1.5 ng/mL as the Cutoff for Prostate Cancer Screening in Primary Care
n this paper, we have presented an alternative approach in which screening is performed for men with at least a 10-year life expectancy. If the PSA is less than 1.5 ng/mL (approximately 70% of men who have a screening PSA), consider a 5-year rescreening interval. If the PSA is ≥ 1.5 ng/mL, or the PCP […]
Association Between a 22-feature Genomic Classifier and Biopsy Gleason Upgrade During Active Surveillance for Prostate Cancer
Insights from this study show that men undergoing active surveillance for their prostate cancer who had a higher Decipher score were more likely to develop aggressive cancer over time. Results conclude that a higher Decipher score might be an indication for tailored active surveillance including frequent prostate biopsies.
Diagnosing hereditary cancer predisposition in men with prostate cancer
This study looks at men with prostate cancer who underwent clinical multigene panel testing. The study found that multigene panel testing as the primary genetic testing approach for hereditary prostate cancer is effective and should be a consideration for germline testing in men with prostate cancer.
Genomic Prostate Score Predicts Recurrence After Radical Prostatectomy, Adverse Surgical Pathology in a Racially Diverse Population of Men
This study of 431 men found that the GPS test is a strong, independent measure of prostate cancer aggressiveness in both African American and Caucasian men as well as an effective test in predicting the recurrence of prostate cancer after a radical prostatectomy.
Genomic Testing in Localized Prostate Cancer Can Identify Subsets of African Americans With Aggressive Disease
This study found the Decipher test to be effective in identifying African American men who are at higher genomic risk for more aggressive prostate cancer. The study concludes that risk stratifying this segment of men can lead to more effective medical interventions earlier in the disease progression.
GPS Assay Association With Long-Term Cancer Outcomes: Twenty-Year Risk of Distant Metastasis and Prostate Cancer–Specific Mortality
This study validates the effectiveness of the Genomic Prostate Score® in predicting distant metastases (DM) and prostate cancer–specific mortality (PCSM) over 20 years.
Hereditary Cancer Risk Assessment and Genetic Testing in the Community-Practice Setting
This study looked at the hereditary cancer risk assessment, education, and testing of patients in the community-based setting and found that genetic testing led to the identification of significant cancer risks that would not have been recognized without the multi-gene panel test.
Identification of men with low-risk biopsy-confirmed prostate cancer as candidates for active surveillance
This study found that the clinical cell-cycle risk (CCR) score threshold is effective in segmenting patients into low- and high-risk groups for 10-year prostate cancer mortality. As a result, the research concludes that CCR (Polaris) may lead to more men being appropriately categorized into active surveillance.
Individual patient-level meta-analysis of the performance of the Decipher Genomic Classifier in high-risk men after prostatectomy to predict development of metastatic disease
This research analyzed five studies of nearly 1,000 men and found the Decipher RP test to be effective in predicting outcomes of patients across all subgroups, regardless of demographics, clinical factors, and treatment approaches.
Prognostic value of a cell cycle progression signature for prostate cancer death in a conservatively managed needle biopsy cohort
This study found that the cell cycle progression (CCP) score (known as Prolaris) may aid in treating men with clinically localized prostate cancer and is the strongest independent predictor of prostate cancer mortality.
Prostate-specific antigen 1.5-4.0 ng/mL: a diagnostic challenge and danger zone
Both Caucasian and African American men with baseline PSA values between 1.5 and 4.0 ng/mL are at increased risk for future prostate cancer compared with those who have an initial PSA value below the 1.5 ng/mL threshold. Based on a growing body of literature and this analysis, it is recommended that a first PSA test threshold of 1.5 ng/mL and above, or somewhere between 1.5 and 4.0 ng/mL, represent the Early-Warning PSA Zone (EWP Zone).
Reduced Rate of Repeated Prostate Biopsies with Confirm mdx
This clinical field utility study of 138 men found that those with negative biopsies who utilized the Confirm mdx test had a significantly lower rate of repeat biopsies.
Validation of a genomic classifier for prediction of metastasis and prostate cancer-specific mortality in African-American men
Like most research, the Decipher genomic classifier test had originally been studied in Caucasian men. This study looked at its accuracy for predicting metastasis and prostate cancer mortality in African American men and found the Decipher test may actually work better in African American men.
Validation of the Decipher genomic classifier in patients receiving salvage radiotherapy without hormone therapy after radical prostatectomy
This study found that patients with a high Decipher genomic classifier score “were more than twice as likely than patients with a lower score to experience biochemical and clinical progression and receive salvage hormone therapy.” Patients with higher scores who were treated early had much better outcomes than those treated later in their disease progression, […]
Validation of the Decipher Test for Predicting Distant Metastatic Recurrence in Men with High-risk Nonmetastatic Prostate Cancer 10 Years After Surgery
This study looked at European and US cases and found the Decipher test is an accurate predictor of distant metastases within 10 years post-surgery.
%s